Research Article - (2021) Volume 11, Issue 2
Development and effectiveness of domestic bait in mouse-like rodents control
A.P. Paliy1*, N.V. Sumakova1, A.A. Antonіuk2, V.L. Behas2 and A.S. Panasenko3Abstract
The fight against synanthropic mouse-like rodents in livestock facilities is of great sanitary, hygienic, and social-economic importance. Today, an effective and scientifically grounded method of rodent control is an integrated approach that ensures the implementation of a full range of veterinary and sanitary measures. The existing chemical agents for combating synanthropic rodents for their improper use can contribute to the formation of rodents that are not sensitive to their action. The work aimed to develop an innovative bait to comb mouse-like rodents and establish its effectiveness in laboratory and production conditions. Based on the results of the studies, formulations of an innovative import-substituting bait for combating rodents were developed: bromadiolone (2.0-2.5%), sunflower oil (2.5-3.0%), sugar (1.0-1.5%), wheat (up to 100%). After giving them bait, the death of white outbred mice was noted within seven days and white outbred rats - within four days by the experiment. When the bait was fed to house mice (Mus musculus) and gray rats (Rattus rattus), their death was noted within 12 and within seven days, respectively. We proved that synanthropic rodents are more resistant to the action of rodenticide bait compared to laboratory animals. The anticoagulant effect of bromadiolone (active ingredient) is manifested by external and internal bleeding in rodents, which leads to their death.
Keywords
bait, recipe, bromadiolone, sunflower oil, sugar, wheat, rats, mice.
Introduction
The earliest companions of humankind and their active competitors in the struggle for existence are mouse-like rodents, common and exist in various climatic zones. In many areas, rodents outnumber all other mammals several times (Morand et al., 2015). Small rodents are increasingly crucial as agricultural pests, although their distribution and abundance differ across landscapes and ecosystems (Fischer & Schröder, 2014). The type of habitat and seasonal changes affect rodents' species composition and relative abundance (Ssuuna et al., 2020). For example, in the Argentine agroecosystems, domestic mice (Mus musculus L., 1758) show a distribution with a large number in livestock farms and a smaller number in crops (León et al., 2010). Other experiments have also proved that Mus musculus most often parasitizes in livestock buildings. Simultaneously, their numbers did not change significantly during the year, while the number of other species is higher in autumn and winter (Miño et al., 2007). It has been proven that, on a long time scale, meteorological variables do not have a direct effect on the number of rodents, but they affect the characteristics of vegetation and are expressed in seasonal variations (Guidobono et al., 2018). Rodents differ from most mammals in their remarkable ecological plasticity (Deacon, 2006; Lutsenko et al., 2014). So, harmful rodents quickly adapt to various conditions of existence; they are characterized by high fertility, polyphagia, good orientation in space, clear organization of behavior in extreme conditions. All this allows them to survive and constantly increase the population size (Pocock et al., 2004; Capdevila et al., 2007). The vital activity of rodents at the expense of food substrates that they receive when parasitizing in livestock farms and complexes, agricultural companies causes significant economic losses, which amount to tens, or even hundreds of thousands of dollars (Stenseth, 2003; Brown et al., 2007; Swanepoel et al., 2017). Mouse-like rodents are gluttonous, which has been proven by laboratory studies. So, the amount of food for a medium-weight rat is about 50 g of grain, equal to 25 kg per year. One mouse eats 3-5 g of grain or 1.1-1.8 kg per year. However, rats and mice spoil foodstuffs significantly more than they eat (Daniels & Hutchings, 2001; Arlet & Molleman, 2007; Mulungu et al., 2007).
In poultry houses, murine rodents can eat up to 20% of the daily diet. The destruction by rats of rabbits, guinea pigs, newborn pigs, and birds has been reported (Stegniy et al., 2012). Rodents destroy not only finished products in barns, warehouses, grocery warehouses, and animal feed but also eat crops of various grain crops, young green shoots, ears of cereals. In the presence of many rodents, they can destroy a significant amount of the crop (Rabiu & Rose, 2004; Brown et al., 2013; Edoh Ognakossan et al., 2018).
The damage done by rodents is not limited to spoilage and destruction of food substrates. So, rodents living in rooms spoil the floor, forming holes and pits, which leads to injury to people and animals. They can cause electrical problems when they chew on wires and cables (Rao & Sakthivel, 2015). In the conditions of industrial livestock complexes, due to the concentration of many livestock in a limited area, the risks of the emergence and spread of infectious diseases of various etiologies increase. To prevent the development of epizootics, it is necessary to control individual chains of the epizootic process. To correctly assess the negative impact of various technological factors on the animal organism, and if a disease occurs, timely diagnose and introduce effective treatment and prevention (Zavgorodniy et al., 2013; Shkromada et al., 2019; Paliy et al., 2020c).
The role of rodents in epidemiological and epizootic aspects is very diverse (Backhans & Fellström, 2012). The main thing is that they are a reservoir and source of infectious diseases in humans and animals, and for a long time retain in their bodies the causative agents of numerous diseases, especially in natural biocenoses (Kilonzo et al., 2013; Gravinatti et al., 2020). The rapid spread of infections is facilitated by relatively high fertility and ubiquitous distribution of rodents, their close contact with each other. More than 50 diseases are known, the causative agents of which can be spread through murine rodents (Rabiee et al., 2018). Thus, rodents are essential reservoirs of pathogens such as leptospirosis, salmonellosis, tularemia, leishmaniasis (Esmaeili et al., 2014; Esfandiari et al., 2017), yersiniosis (Backhans et al., 2011), toxoplasmosis (Paknejadi et al., 2002), plague, hantavirus infections (Enria & Pinheiro, 2000). Escherichia coli (10 isolates), Salmonella typhimurium (7 isolates), Shigella sonnei (1 isolate), Proteus mirabilis (3 isolates), Citrobacter freundii (17 isolates), and Enterobacter (4 isolates) were isolated from rats, which had increased resistance to antibacterial drugs (Najar Peerayeh et al., 2010).
In the conditions of Ukraine, among synanthropic rodents that cause significant harm to livestock, the most common are rats, mice, and voles. They often live in dwellings, livestock buildings, and grazing areas for farm animals (Stegniy et al., 2012). Rodent control is receiving much attention in many countries worldwide (Suárez & Cueto, 2018). In the USA alone, more than 80 different companies are involved in producing and selling rodenticides. About 1300 different rodent-killing bait recipes, some with up to 30 different ingredients (Parker & Townsley, 2004; Sayer, 2017). Simultaneously, existing chemical agents for the control of synanthropic rodents can contribute to the formation of resistance to a particular active substance (Hodroge et al., 2011; Song et al., 2011). Thus, the frequent use of anticoagulants and the constant presence of M. musculus and Rattus norvegicus indicate the presence of these rodents' resistance populations (Meerburg et al., 2014; León et al., 2020).
For the successful organization of effective control of murine rodents, it is necessary to know the features of their biological development and behavior, the relationship of synanthropic rodents with natural ecosystems, their migration routes (Mari Saez et al., 2018; Mariën et al., 2019). To control harmful rodents, their natural enemies are used (Paz et al., 2013; Adduci et al., 2021). Daily capture and destruction of harmful rodents in places of their greatest concentration are considered effective (Taylor et al., 2012). The actual harm caused by rodents to the livestock industry is difficult to reflect in monetary terms, so the fight against them should be systematic and scientifically based.
Materials and Methods
The work aimed to develop a bait for controlling the mouse-like rodents and verify its effectiveness in a laboratory and industrial conditions. Experimental studies were carried out in the laboratory of veterinary sanitation and parasitology of the National Scientific Center "Institute of Experimental and Clinical Veterinary Medicine" (Kharkiv).
Bromadiolone (active ingredient), sunflower oil (food attractant), sugar (food attractant), wheat (nutrient base) were used to design the formulation of the innovative bait at various ratios in the bait. The mass fraction of bromadiolone was measured by high-performance liquid chromatography using a UV detector, chromatographic separation in isocratic mode, and quantitative assessment by absolute calibration. At the preliminary stage, preparation for the control was carried out:
1.Preparation of the mobile phase (eluent) – in a cylinder with a capacity of 100 cm3, 80 cm3 of acetonitrile, and 20 cm3 of water were mixed, after which the mixture was degassed.
2.Preparation of the primary calibration solution of bromadiolone – 0.04 g of bromadiolone was dissolved in acetonitrile in a volumetric flask with a capacity of 100 cm3; the volume was brought to the mark and stirred.
3.Preparation of a working calibration solution of bromadiolone – 0.3 cm3 of the main calibration solution was dosed into a 25 cm3 volumetric flask, and the eluent was added to the mark. After stirring, the solution was chromatographed, and from the obtained chromatograph, the chromatographic peak area was determined with bromadiolone.
Chromatography parameters: wavelength (254 nm), eluent velocity (0.5 cm3/min.), column temperature (37°С), dose-volume (20 μl). The retention time for bromadiolone was approximately 5.5 minutes. In a volumetric flask with a capacity of 50 cm3, 0.1 g of the bait was dissolved in acetonitrile, the eluent was added to the mark, and, after stirring, it was dosed into the chromatography using an injector. The mass fraction of bromadiolone was calculated by the formula:
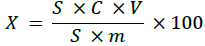
S – the area of the chromatographic peak of bromadiolone in the test (working graduated) solution; C – the concentration of bromadiolone in the working grаduated solution, mg /cm3;
V – the volume of the test solution, cm3; m – sample mass, mg;
100 – the coefficient.
Data processing was carried out following the current recommendations (Zuur et al., 2010). Determination of the effectiveness of the developed bait was carried out according to the scheme:
1)the formation of research and control groups of animals;
2)the use of bait, keeping animals, determining the effectiveness of the bait;
3)daily clinical examination of experimental animals during the entire experiment;
4)postmortem examination of dead animals.
To develop a bait for combating murine rodents, the active ingredient was used – bromadiolone, food attractants (sugar, sunflower oil), and a nutritional base (wheat). Bromodialon is a second-generation rodenticide of 4-hydroxycoumarin derivatives. In case of acute toxicity with potentially dangerous routes of entry into the body, bromadiolone belongs to class 1 of highly hazardous substances for oral and inhalation exposure in the form of an aerosol and class 2 of highly hazardous substances when applied to the skin. There is insufficient information on bromadiolone's toxicodynamics and toxicokinetics in humans, and further research is needed (Lo et al., 2008).
To determine the most rational composition of domestic bait against mouse-like rodents, three of its formulations were studied (Table 1).
| No | The composition of the bait | Recipe, wt % | ||
|---|---|---|---|---|
| І | ІІ | ІІІ | ||
| 1 | Bromodialon | 2.0 | 2.125 | 2.5 |
| 2 | Sunflower oil | 2.0 | 2.5 | 3.0 |
| 3 | Sugar | 1.0 | 1.5 | 1.5 |
| 4 | Wheat | up to 100 | up to 100 | up to 100 |
Table 1. Recipe for the bait for the control of mouse rodents
Tests of the action of the components of the bait were carried out on laboratory white outbred mice (n=40), white outbred rats (n=40), and house mice (Mus musculus) (n=40) and gray rats (Rattus rattus) (n=24). Rodent body weight: laboratory white mice – 25.0±0.5 g, house mice – 15.5-26.0 g, laboratory rats – 300.0±0.5 g, gray rats – 280.5-335.5 g. Research mice were fed baits with different formulations at a dose of 5 g per animal and rats at 15 g. Control animals were given pure grain. Rodents received bait daily throughout the experiment. Simultaneously with the bait, the rodents were getting plenty of water. During the experiment, observations were made for the development of clinical signs of poisoning in rodents. The animals that died during the experiment were subjected to postmortem autopsy to detect lesions of internal organs (Kotsiumbas et al., 2006). Experimental studies on animals were carried out considering the basic principles of bioethics (Farnfield et al., 2017; Kabene & Baadel, 2019).
Results
As a result of the analysis of literature sources and patent search, it was found that the disadvantages of existing baits for combating mouse-like rodents are their low biological activity, poor eating ability, and unattractiveness for rodents. Rodents have developed resistance to many toxic substances, due to which they are ineffective (Tanaka et al., 2012; Marquez et al., 2019). The most effective means in the fight against mouse-like rodents are anticoagulants' use (Frankova et al., 2019).
We have identified the most promising constituent components for designing a domestic rodenticide agent (Table 2).
| No | Сomponent | Wt % |
|---|---|---|
| 1 | Bromodialon (active ingredient) | 2.0-2.5 |
| 2 | Sunflower oil (food attractant) | 2.5-3.0 |
| 3 | Sugar (food attractant) | 1.0-1.5 |
| 4 | Wheat (nutrient base) | up to 100 |
Table 2. The composition of the bait for the control of murine rodents
Bromadiolone is a second generation coumarin anticoagulant. It acts on the synthesis of prothrombin and permeability of blood vessels, leading to a disruption of the blood coagulation mechanism, causing slow death from bleeding (Pelz et al., 2005). To attract rodents to the bait, fragrant sunflower oil (2.5-3.0%) is added. To better eat the bait, a food attractant – sugar (1.0- 1.5%) was introduced into its composition, and cereals (wheat) were selected as a nutritional base.
To determine the most rational composition of the domestic bait against mouse-like rodents, its various formulations were studied for actions on laboratory animals. For this purpose, three experimental and one control group of white outbred mice and white outbred rats were formed separately. Experimental animals of groups No. 1 were fed the bait according to the recipe I, the animals of groups No. 2 were given the bait according to the recipe of II, and the animals of groups No. 3 were given the agent according to the recipe of III. Control animals were given pure grain (Table 3).
| Group | Number ofanimals | Death of rodents, days | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | ||
| Mice | |||||||||||||
| No 1 | 10 | – | – | 2 | 2 | 3 | 2 | 1 | – | – | – | – | – |
| No 2 | 10 | – | – | 2 | 3 | 5 | – | – | – | – | – | – | – |
| No 3 | 10 | – | 3 | 7 | – | – | – | – | – | – | – | – | – |
| control | 10 | – | – | – | – | – | – | – | – | – | – | – | – |
| Rats | |||||||||||||
| No 1 | 10 | – | 1 | 4 | 5 | – | – | – | – | – | – | – | – |
| No 2 | 10 | – | 2 | 4 | 4 | – | – | – | – | – | – | – | – |
| No 3 | 10 | – | 4 | 6 | – | – | – | – | – | – | – | – | – |
| control | 10 | – | – | – | – | – | – | – | – | – | – | – | – |
Table 3. The action of a rodenticidal agent on laboratory animals
According to the research results, it was found that the rodenticidal agent in the composition of bromadiolone – 2.0%, sunflower oil – 2.0%, sugar – 1.0%, wheat – up to 100% (a recipe I) causes the death of all mice on the 7th day after eating. It has also been proven that a bait that contains bromadiolone – 2.125%, sunflower oil – 2.5%, sugar – 1.5%, wheat – up to 100% (recipe II) causes the death of 100% of mice on the 5th day after application, and the bait with the ratio of the components bromadiolone – 2.5%, sunflower oil – 3.0%, sugar – 1.5%, wheat – up to 100% (recipe III) acts lethally (100%) on laboratory mice on the 3rd day after feeding. It should be noted that rodents more readily ate the bait prepared according to recipe III. The first clinical manifestations of poisoning were observed 6 hours after applying the bait on the same day. In rats, when feeding the bait I and
IIformulations, death occurred from 2 to 4 days, and formulations III – on the second and third days of the experiment with characteristic pathological changes in internal organs at dissection.
At the next stage of research, the effect of the developed bait on synanthropic rodents was determined. For this purpose, research and control groups of animals were formed, and different formulations of the bait were assigned following the previous experiment (Table 4).
| Group | Number ofanimals | Death of rodents, days | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | ||
| Mus musculus | |||||||||||||
| No 1 | 10 | – | – | – | – | – | 1 | 2 | 2 | 1 | 2 | 1 | 1 |
| No 2 | 10 | – | – | – | – | 1 | – | 2 | 2 | 2 | 1 | 2 | |
| No 3 | 10 | – | – | – | – | – | 1 | 2 | 3 | 1 | 1 | 1 | 1 |
| control | 10 | – | – | – | – | – | – | – | – | – | – | – | – |
| Rattus rattus | |||||||||||||
| No 1 | 6 | – | – | – | 1 | 2 | 2 | 1 | – | – | – | – | – |
| No 2 | 6 | – | – | – | 2 | 1 | 2 | 1 | – | – | – | – | – |
| No 3 | 6 | – | – | – | 1 | 2 | 1 | 2 | – | – | – | – | – |
| control | 6 | – | – | – | – | – | – | – | – | – | – | – | – |
Table 4. The effect of the bait on house mice (Mus musculus) and gray rats (Rattus rattus)
Among synanthropic rodents, the first clinical manifestations of poisoning were observed in animals after 48 hours. The animals refused to eat the bait; there was thirst, bloody discharge from the eyes. After 72 hours, bloody discharge from the nasal cavity appeared; in many animals, the forelegs' hair was contaminated with blood. From the fourth day of the experiment, the death of the experimental rats began to be recorded, and from the fifth day, the death of research mice. However, it was not widespread among house mice, and the last mouse died on the 12th day. Among gray rats, animals' death was noted from the fourth to the seventh day; in the latter, the smallest rat by weight died. In the control groups, all animals were alive and active. So, synanthropic rodents are more resistant to the action of the bait than laboratory animals. It was also noted that rodents eat bait with food attractants better than untreated grain (control). On external examination of the rodents, it was noted that rigor mortis was poorly expressed, the visible mucous membranes were pale, and the skin was bluish in the head and abdomen region.
Postmortem examination of mice revealed extensive hemorrhages in the subcutaneous tissue. The blood is liquid, bright red, watery. In the chest cavity, there is a cloudy red serous fluid. On the lungs, heart, liver marked multiple hemorrhages. The stomach is swollen, the contents are liquid, bloody, the gastric mucosa is hyperemic. In the small part of the intestine, the contents are yellow, with a viscous consistency. The intestinal mucosa is swollen, thickened, yellow gelatinous infiltrates are visible on the mesentery. In the large intestine, the contents are liquid, viscous, with blood impurities. In rats, rigor mortis is weak. The skin is bluish; in some places, there are wounds with blood on the skin. At autopsy, hemorrhages were noted in the subcutaneous tissue, abdomen, chest. The gastric mucosa is pink. The liver is yellowish to dark red, loose.
Spleen without visible changes. The kidneys are unevenly colored, from light brown to brown. Across the entire surface of the kidneys, there are punctate hemorrhages. The small intestine is filled with yellow viscous contents. In each lobe of the lung, punctate hemorrhages are noted. The vessels of the heart are filled with blood. Pathological studies of organs have shown that when the bait's active substance (bromadiolone) enters the animal's body, blood clotting is disturbed, the walls of peripheral vessels are damaged, hemorrhagic diathesis, internal hemorrhages, and pulmonary edema develop, which, in turn, is lethal. Thus, bromadiolone causes both external and internal bleeding in rodents due to its anticoagulant action. Thus, bromadiolone causes both external and internal bleeding in rodents' bodies due to its anticoagulant effect.
In production conditions, the bait's effectiveness was tested on the experimental base of the NSC "IEKVM". In warehouses along the perimeter, 25 g of pre-bait was laid out every 5 m. The amount of bait was replenished every day as rodents ate it. The first nine corpses of rats in the room were found on the 5th day after applying the bait; on the sixth and seven days, 7 and 5 rats were detected, respectively. On the sixth day, the first dead mice were found in several rooms; the bait was eaten and replenished for up to 8 days, the last corpses of mice were collected on the tenth day. In total, during the deratization, 50 corpses of mice and 24 corpses of rats were collected. Among the dead rodents were adult males and females, young animals of different ages. The results obtained confirm the rodenticidal effect of the developed domestic import-substituting bait for combating murine rodents. Based on the research results, a patent for a rodenticidal agent was obtained (Paliy et al., 2020a).
Discussion
The modern complex of veterinary, sanitary, and technological measures at agricultural enterprises provides for the implementation of disinfection (Paliy et al., 2018a; Kovalenko et al., 2020), disinsection (George et al., 2008; Paliy et al., 2018d), deacarization (Walker 2011; Paliy et al., 2018b), disinfestation (Melnychuk & Yuskiv, 2018; Paliy et al., 2020b) and deworming (Shrestha et al., 2020; Paliy et al., 2021), due to the ubiquitous spread of pathogens of various diseases animals (Miller et al., 2017; Delahoy et al., 2018; Paliy et al., 2018c, 2019). Along with this, an essential component of the control of the veterinary and sanitary state of any livestock enterprise is the systematic and planned control of mouse-like rodents (Janković et al., 2019).
To control rodents, it is necessary to use highly effective rodenticides, the effectiveness of which has been confirmed both in the laboratory and industrial conditions (Buckle et al., 2012; Goulois et al., 2017). The relevance and need for constant control of murine rodents have led to the development and use of various rodenticide agents of various origins. Studies of the effectiveness of some different baits are 100% effective regardless of the type of formulation and manufacturer (Suárez & Cueto, 2018). The inhibitory effect of Chlorpyrifos on the rat organism was studied (Rosalovsky et al., 2015). In our experiments, we used bromadiolone and brought its highly rodenticidal action regardless of the length of its use directly in production. Other researchers have shown that 0.005% bromadiolone completely eradicates R. norvegicus and R. rattus in 1 and 5 days, respectively, and M. musculus in 12 days (Redfern & Gill, 1980). However, information on the potential risk of an increase in the proportion of resistant individuals at the genetic level and a decrease in bromadiolone effectiveness with prolonged and uncontrolled use should be taken into account (León et al., 2020).
Our results agree with those regarding innovative, environmentally friendly baits (Horak et al., 2018). However, in the field, control with artificial baits is not very effective since rodents have various alternative food (Guidobono et al., 2010; Adduci et al., 2019).
A proven fact in rodent control, the need for careful planning of field hygiene control measures, where the fields must be free of weeds, in turn, reduces the potential breeding and existence of rodents, thereby reducing the rate of their recovery and population growth (Mayamba et al., 2020). Rodent control programs should achieve better results if health agencies focus on areas with more volatile conditions and industrial areas during colder seasons when rodent numbers are less (Cavia et al., 2015). Regular screening of rodents' specific characteristics and the environmental conditions of specific agricultural enterprises make it possible to successfully control the number of rodents and reduce losses from their harmful activities.
Conclusions
A formulation of an innovative import-substituting bait for combating mouse-like rodents has been developed, containing bromadiolone (2.0-2.5%) as an active ingredient, as well as sunflower oil (food attractant) (2.5-3.0%), sugar (food attractant) (1.0- 1.5%), wheat (nutritional base) (up to 100%). The effectiveness of the developed charm has been proven on laboratory and synanthropic rodents. For actions on white outbred mice, their death was noted from 2 to 7 days, and white outbred rats - from 2 to 4 days of the experiment. When food was fed to house mice (Mus musculus) and gray rats (Rattus rattus), their deaths were noted from 5 to 12 and from 4 to 7 days, respectively. During postmortem autopsy of animal corpses after feeding the bait, external and internal bleeding was noted due to the bromadiolone anticoagulant effect. We proved that synanthropic rodents are more resistant to rodenticide bait action than laboratory animals. The rodenticide effect of the developed bait for combating mouse rodents has been confirmed in production conditions, which gives rise to its widespread introduction into production. The results of the conducted research expand the range of products for pest control in agricultural production.
References
Adduci, L. B., León, V. A., Busch, M., & Fraschina, J. (2019). Effects of different odours on the reproductive success of Mus musculus as an alternative method of control. Pest Manag Sci., 75(7), 1887-1893. doi: 10.1002/ps.5359
Adduci, L. B., León, V. A., Schlötelburg, A., Busch, M., & Fraschina, J. (2021). Avoidance behaviour in laboratory house mice (Mus musculus) and Norway rats (Rattus norvegicus) towards predator odours. PLOS ONE, 16, 1, e0245441. doi: 10.1371/journal.pone.0245441
Arlet, M. E., & Molleman, F. (2007). Rodents damage crops more than wildlife in subsistence agriculture on the northern periphery of Dja Reserve, Cameroon. International Journal of Pest Management, 53(3), 237-243. doi: 10.1080/09670870701418994
Backhans, A., Fellström, C., & Lambertz, S. T. (2011). Occurrence of pathogenic Yersinia enterocolitica and Yersinia pseudotuberculosis in small wild rodents. Epidemiol Infect., 139, 1230-1238.
Backhans, A., & Fellström, C. (2012). Rodents on pig and chicken farms – a potential threat to human and animal health. Infection Ecology & Epidemiology, 2, 1. doi: 10.3402/iee.v2i0.17093
Brown, P., Huth, N. I., Banks, P. B., & Singleton, G. R. (2007). Relationship between abudance of rodents and damage to agricultural crops. Agriculture Ecosystems & Environment, 120(2), 405-415. doi: 10.1016/j.agee.2006.10.016
Brown, P., McWilliam, A., & Khamphoukeo, K. (2013). Post-harvest damage to stored grain by rodents in village environments in Laos. International Biodeterioration & Biodegradation, 82, 104-109. doi: 10.1016/j.ibiod.2012.12.018
Buckle, A. P., Klemann, N., & Prescott, C. V. (2012). Brodifacoum is effective against Norway rats (Rattus norvegicus) in a tyrosine139cysteine focus of anticoagulant resistance in Westphalia, Germany. Pest. Manag. Sci., 68(12), 1579-1585. doi: 10.1002/ps.3352
Capdevila, S., Giral, M., de la Torre, J. L. R., Russell, R. J., & Kramer, K. (2007). Acclimatization of rats after ground transportation to a new animal facility. Laboratory Animals, 41, 255-261.
Cavia, R., Muschetto, E., Cueto, G. R., & Suárez, O. V. (2015). Commensal Rodents in the City of Buenos Aires: A Temporal, Spatial, and Environmental Analysis at the Whole City Level. Ecohealth., 12(3), 468-479. doi: 10.1007/s10393-015-1013-8
Daniels, M. J., & Hutchings, M. R. (2001). The response of cattle and sheep to feed contaminated with rodent faeces. Vet J., 162(3), 211-218. doi: 10.1053/tvjl.2000.0552
Deacon, R. M. (2006). Housing, husbandry and handling of rodents for behavioral experiments. Nat Protoc., 1(2), 936-946. doi: 10.1038/nprot.2006.120
Delahoy, M. J., Wodnik, B., McAliley, L., Penakalapati, G., Swarthout, J., Freeman, M. C., & Levy, K. (2018). Pathogens transmitted in animal feces in low- and middle- income countries. Int J Hyg Environ Health., 221(4), 661-676. doi: 10.1016/j.ijheh.2018.03.005
Edoh Ognakossan, K., Mutungi, C. M., Otieno, T. O., Affognon, H. D., Sila, D. N., & Owino, W. O. (2018). Quantitative and quality losses caused by rodents in on-farm stored maize: a case study in the low land tropical zone of Kenya. Food Sec., 10, 1525-1537. doi: 10.1007/s12571-018-0861-9
Enria, D. A., & Pinheiro, F. (2000). Rodent-borne emerging viral zoonosis. Hemorrhagic fevers and hantavirus infections in South America. Infect Dis Clin North Am., 14(1), 167-184. doi: 10.1016/s0891-5520(05)70223-3
Esfandiari, B., Nahrevanian, H., Pourshafie, M. R., Gouya, M. M., Khaki, P., Mostafavi, E., Darvish, J., & Hanifi, H. (2017). Epidemiological Distribution of Rodents as Potent Reservoirs for Infectious Diseases in the Provinces of Mazandaran, Gilan and Golestan, Northern Iran. Infectious disease reports, 9(2), 6900. doi: 10.4081/idr.2017.6900
Esmaeili, S., Gooya, M. M., Shirzadi, M. R., Esfandiari, B., Amiri, F. B., Behzadi, M. Y., Banafshi, O., & Mostafavi, E. (2014). Seroepidemiological survey of tularemia among different groups in western Iran. Int J Infect Dis., 18, 27-31. doi: 10.1016/j.ijid.2013.08.013
Farnfield, B., Petrie, A., & Dennison, N. (2017). Use of animals in research. Vet Rec., 181(7), 178-179. doi: 10.1136/vr.j3828
Fischer, C., & Schröder, B. (2014). Agriculture, ecosystems and environment predicting spatial and temporal habitat use of rodents in a highly intensive agricultural area. Agric. Ecosyst. Environ., 189, 145-153. doi: 10.1016/j.agee.2014.03.039
Frankova, M., Stejskal, V., & Aulicky, R. (2019). Efficacy of rodenticide baits with decreased concentrations of brodifacoum: Validation of the impact of the new EU anticoagulant regulation. Sci Rep., 9, 16779(2019). doi: 10.1038/s41598-019-53299-8
George, D. R., Guy, J. H., Arkle, S., Harrington, D., De Luna, C., Okello, E. J., Shiel, R. S., Port, G., & Sparagano, O. A. (2008). Use of plant-derived products to control arthropods of veterinary importance: a review. Ann N Y Acad Sci., 1149, 23-26. doi: 10.1196/annals.1428.021
Goulois, J., Hascoët, C., Besse, S., Legros, L., Benoit, E., & Lattard, V. (2017). Study of the efficiency of anticoagulant rodenticides to control Mus musculus domesticus introgressed with Mus spretus Vkorc1. Pest Management Science, 73(2), 325-331. doi: 10.1002/ps.4319
Gravinatti, M. L., Barbosa, C. M., Soares, R. M., & Gregori, F. (2020). Synanthropic rodents as virus reservoirs and transmitters. Journal of the Brazilian Society of Tropical Medicine, 53, e20190486. doi: 10.1590/0037-8682-0486-2019
Guidobono, J. S., Cueto, G. R., Teta P., & Busch, M. (2018). Effect of environmental factors on the abundance variations of two native rodents in agricultural systems of Buenos Aires, Argentina. Austral Ecol., 44, 36-48. doi: 10.1111/aec.12650
Guidobono, J. S., León, V., Gómez Villafañe, I. E., & Busch, M. (2010). Bromadiolone susceptibility in wild and laboratory Mus musculus L. (house mice) in Buenos Aires, Argentina. Pest Management Science, 66(2), 162-167. doi: 10.1002/ps.1850
Hodroge, A., Longin-Sauvageon, C., Fourel, I., Benoit, E., & Lattard, V. (2001). Biochemical characterization of spontaneous mutants of rat VKORC1 involved in the resistance to antivitamin K anticoagulants. Arch. Biochem. Biophys., 515(1-2), 14-20. doi: 10.1016/j.abb.2011.08.010
Horak, K., Fisher, P. M., & Hopkins, B. M. (2018). Pharmacokinetics of Anticoagulant Rodenticides in Target and Nontarget Organisms. USDA National Wildlife Research Center - Staff Publications. 2091. https://digitalcommons.unl.edu/icwdm_usdanwrc/2091
Janković, L., Drašković, V., Pintarič, Š., Mirilović, M., Đurić, S., Tajdić, N., & Teodorović, R. (2019). Rodent pest control. Veterinarski Glasnik, 73(2), 85-99. doi: 10.2298/VETGL190507020J
Kabene, S., & Baadel, S. (2019). Bioethics: a look at animal testing in medicine and cosmetics in the UK. Journal of medical ethics and history of medicine, 12, 15. doi: 10.18502/jmehm.v12i15.1875
Kilonzo, C., Li, X., Vivas, E. J., Jay-Russell, M. T., Fernandez, K. L., & Atwill, E. R. (2013). Fecal shedding of zoonotic food-borne pathogens by wild rodents in a major agricultural region of the central California coast. Applied and environmental microbiology, 79(20), 6337-6344. doi: 10.1128/AEM.01503-13
Kotsiumbas, I. Ya., Malik, O. G. & Paterega, I. P. (2006). Preclinical studies of veterinary drugs: Lviv: Triada plus. (in Ukrainian)
Kovalenko, V. L., Ponomarenko, G. V., Kukhtyn, M. D., Paliy, A. P., Bodnar, O. O., Rebenko, H. I., Kozytska, T. G., Makarevich, T. V., Ponomarenko, O. V., & Palii, A. P. (2020). Evaluation of acute toxicity of the "Orgasept" disinfectant. Ukrainian Journal of Ecology, 10(4), 273-278. doi: 10.15421/2020_199
León, V. A., Fraschina, J., & Busch, M. (2020). Bromadiolone susceptibility in Mus musculus (house mice) of Argentina. International Journal of Pest Management, 66, 1, 7-12. doi: 10.1080/09670874.2018.1538543
León, V. A., Fraschina, J., & Busch, M. (2010). Population subdivision of house mice (Mus musculus) in an agrarian landscape: consequences for control. Canadian Journal of Zoology, 88, 427-435. doi: 10.1139/Z10-014
Lo, V. M., Ching, C. K., Chan, A. Y., & Mak, T. W. (2008). Bromadiolone toxicokinetics: diagnosis and treatment implications. Clin Toxicol (Phila)., 46(8), 703-710. doi: 10.1080/15563650701504366
Lutsenko, D. G., Shylo, O. V., Sleta, I. V., & Marchenko, V. S. (2014). Peculiarities of rat brain microhemocirculation after various modes of long-term cold-acclimation. Neurophysiology, 46(4), 384-386. doi: 10.1007/s11062-014-9460-3
Mariën, J., Borremans, B., Kourouma, F., Baforday, J., Rieger, T., Günther, S., Magassouba, N., Leirs, H., & Fichet-Calvet, E. (2019). Evaluation of rodent control to fight Lassa fever based on field data and mathematical modeling. Emerging Microbes & Infections, 8, 1, 640-649. doi: 10.1080/22221751.2019.1605846
Mari Saez, A., Cherif Haidara, M., Camara, A., Kourouma, F., Sage, M., Magassouba, N., & Fichet-Calvet, E. (2018). Rodent control to fight Lassa fever: Evaluation and lessons learned from a 4-year study in Upper Guinea. PLoS neglected tropical diseases, 12(11), e0006829. doi. 10.1371/journal.pntd.0006829
Marquez, A., Khalil, R. A., Fourel, I., Ovarbury, T., Pinot, A., Rosine, A., Thalmensi, G., Jaffory, G., Kodjo, A., Benoit, E., & Lattard, V. (2019). Resistance to anticoagulant rodenticides in Martinique could lead to inefficient rodent control in a context of endemic leptospirosis. Sci Rep., 9, 13491(2019). doi: 10.1038/s41598-019- 49661-5
Mayamba, A., Byamungu, R. M., Broecke, B. V., Leirs, H., Hieronimo, P., Nakiyemba, A., Isabirye, M., Kifumba, D., Kimaro, D. N., Mdangi, M. E., & Mulungu, L. S. (2020). Factors influencing the distribution and abundance of small rodent pest species in agricultural landscapes in Eastern Uganda. Journal of Vertebrate Biology, 69(2), 20002, 1-17. doi: 10.25225/jvb.20002
Meerburg, B. G., van Gent-Pelzer, M. P., Schoelitsz, B. & Van der Lee, T. A. (2014). Distribution of anticoagulant rodenticide resistance in Rattus norvegicus in the Netherlands according to Vkorc1 mutations. Pest. Manag. Sci., 70(11), 1761-1766. doi: 10.1002/ps.3809
Melnychuk, V., & Yuskiv, I. (2018). Disinvasive efficacy of chlorine-based preparations of domestic production for eggs of nematodes of the species Aonchotheca bovis parasitizing in sheep. Ukrainian Journal of Veterinary and Agricultural Sciences, 1(2), 15-18. doi: 10.32718/ujvas1-2.04
Miller, R. S., Sweeney, S. J., Slootmaker, C., Grear, D. A., Di Salvo, P. A., Kiser, D., & Shwiff, S. A. (2017). Cross-species transmission potential between wild pigs, livestock, poultry, wildlife, and humans: implications for disease risk management in North America. Scientific Reports, 7, 7821(2017). doi: 10.1038/s41598- 017-07336-z
Miño, M. H., Cavia, R., Gómez Villafañe, I. E., Bilenca, D. N., & Busch, M. (2007). Seasonal abundance and distribution among habitats of small rodents on poultry farms. A contribution for their control. International Journal of Pest Management, 12, 1-6. doi: 10.1080/09670870601105949
Morand, S., Bordes, F., Blasdell, K., Pilosof, S., Cornu, J.-F., Chaisiri, K., Chaval, Y., Cosson, J.-F., & Claude, J. (2015). Assessing the distribution of disease‐bearing rodents in human‐modified tropical landscapes. Journal of Applied Ecology, 52(3), 784-794. doi: 10.1111/1365-2664.12414
Mulungu, L. S., Makundi, R. H., Massawe, A. W., & Leirs, H. (2007). Relationship between sampling intensity and precision for estimating damage to maize caused by rodents. Integr Zool., 2(3), 131-135. doi: 10.1111/j.1749-4877.2007.00051.x
Najar Peerayeh, S., Soleimani, N., Sadrai, J., & Derakhshan, S. (2010). Investigation of Contamination of Wild Rats (Rattus rattus) from Tehran City to Antibiotic Resistant Enterobacteriaceae in 2009. J Mazandaran Univ Med Sci., 20(78), 70-75. (in Persian)
Paknejadi, M., Asmar, M., & Amirjani, A. (2002). Toxoplasma gondii Infection of Stored Rodents in Lahijan. Iran J Infect Dis Trop Med., 6(14), 56.
Parker, J. C., & Townsley, M. I. (2004). Evaluation of lung injury in rats and mice. Am J Physiol Lung Cell Mol Physiol., 286(2), 231-246. doi: 10.1152/ajplung.00049.2003
Paliy, A. P., Ishchenko, K. V., Marchenko, M. V., Paliy, A. P., & Dubin, R. A. (2018a). Effectiveness of aldehyde disinfectant against the causative agents of tuberculosis in domestic animals and birds. Ukrainian Journal of Ecology, 8(1), 845-850. doi: 10.15421/2018_283
Paliy, A. P., Mashkey, A. M., Sumakova, N. V., & Paliy, A. P. (2018b). Distribution of poultry ectoparasites in industrial farms, farms, and private plots with different rearing technologies. Biosystems Diversity, 26(2), 153-159. doi: 10.15421/011824
Paliy, A. P., Petrov R. V., Kovalenko, L. M., Livoshchenko, L. P., Livoshchenko, Y. M., Klishchova, Z. E., Bula, L. V., Ostapenko, V. I., Doletskyi, S. P., & Palii, A. P. (2021). Effectiveness of a modern antiparasitic agent for deworming in domestic animals. Ukrainian Journal of Ecology, 11(1), 11-17. doi: 10.15421/2020_302
Paliy, A. P., Sumakova, N. V., Mashkey, A. M., Petrov, R. V., Paliy, A. P., & Ishchenko, K. V. (2018c). Contamination of animal-keeping premises with eggs of parasitic worms. Biosystems Diversity, 26(4), 327-333. doi: 10.15421/011849
Paliy, A. P., Sumakova, N. V., Paliy, A. P., & Ishchenko, K. V. (2018d). Biological control of house fly. Ukrainian Journal of Ecology, 8(2), 230-234. doi:10.15421/2018_332 Paliy, A. P., Mashkey, A. M., Sumakova, N. V., & Stegniy, B. T. (2020a). Patent № 142938 UA, МPK А01M 19/00; А01N 25/00. Bait to combat rodents "Ratlon" / applicant
and patentee NSC "IEСVM". - № u 2019 10781; pend. 31.10.19; publ. 10.07.20, Bul. № 13, p. 3.
Paliy, A. P., Sumakova, N. V., Rodionova, K. O., Nalivayko, L. I., Boyko, V. S., Ihnatieva, T. M., Zhigalova, O. Ye., Dudus, T. V., Anforova, M. V., & Kazakov, M. V. (2020b). Disinvasive action of aldehyde and chlorine disinfectants on the test-culture of Toxocara canis eggs. Ukrainian Journal of Ecology, 10(4), 175-183. doi: 10.15421/2020_185
Paliy, A. P., Zavgorodniy, A. I., Stegniy, B. T., & Palii, A. P. (2020c). Scientific and methodological grounds for controlling the development and use of disinfectants. Monograph. Kharkiv: «Miskdruk», 318. ISBN: 978-617-619-237-4. (in Ukrainian)
Paliy, A., Sumakova, N., Petrov, R., Shkromada, O., Ulko, L., & Palii, A. (2019). Contamination of urbanized territories with eggs of helmiths of animals. Biosystems Diversity, 27(2), 118-124. doi: 10.15421/011916
Paz, A., Jareño, D., Arroyo, L., Viñuela, J., Arroyo, B., Mougeot, F., Luque-Larena, J. J., & Fargallo, J. A. (2013). Avian predators as a biological control system of common vole (Microtus arvalis) populations in north-western Spain: experimental set-up and preliminary results. Pest Manag Sci., 69(3), 444-450. doi: 10.1002/ps.3289
Pelz, H. J., Rost, S., Hünerberg, M., Fregin, A., Heiberg, A. C., Baert, K., MacNicoll, A. D., Prescott, C. V., Walker, A. S., Oldenburg, J., & Müller, C. R. (2005). The genetic basis of resistance to anticoagulants in rodents. Genetics, 170(4), 1839-1847. doi: 10.1534/genetics.104.040360
Pocock, M. J. O., Searle, J. B., & White, P. C. L. (2004). Adaptations of animals to commensal habitats: population dynamics of house mice Mus musculus domesticus on farms. Journal of Animal Ecology, 73(5), 878-888. doi: 10.1111/j.0021-8790.2004.00863.x
Rabiee, M. H., Mahmoudi, A., Siahsarvie, R., Kryštufek, B., & Mostafavi, E. (2018). Rodent-borne diseases and their public health importance in Iran. PLoS neglected tropical diseases, 12(4), e0006256. doi: 10.1371/journal.pntd.0006256
Rabiu, S., & Rose, R. K. (2004). Crop damage and yield loss caused by two species of rodents in irrigated fields in northern Nigeria. International Journal of Pest Management, 50, 4, 323-326. doi: 10.1080/09670870400000374
Rao, A. M. K. M., & Sakthivel, P. (2015). Role of rodents in poultry environs and their management. J Dairy Vet Anim Res., 2(3), 107-114. doi: 10.15406/jdvar.2015.02.00040
Redfern, R., & Gill, J. E. (1980). Laboratory evaluation of bromadiolone as a rodenticide for use against warfarin-resistant and non-resistant rats and mice. J Hyg (Lond)., 84(2), 263-268. doi: 10.1017/s0022172400026760
Rosalovsky, V. P., Grabovska, S. V., & Salyha, Yu. T. (2015). Changes in glutathione system and lipid peroxidation in rat blood during the first hour after chlorpyrifos exposure. Ukr. Biochem. J., 87(5), 124-132. doi: 10.15407/ubj87.05.124
Sayer, K. (2017). The 'modern' management of rats: British agricultural science in farm and field during the twentieth century. BJHS Themes, 2, 235-263. doi:10.1017/bjt.2017.7
Shkromada, O., Skliar, O., Paliy, A., Ulko, L., Gerun, I., Naumenko, О., Ishchenko, K., Kysterna, O., Musiienko, O., & Paliy, A. (2019). Development of measures to improve milk quality and safety during production. Eastern-European Journal of enterprise technologies, 3/11(99), 30-39. doi: 10.15587/1729- 4061.2019.168762
Shrestha, U. T., Adhikari, N., Kafle, S., Shrestha, N., Banjara, M. R., Steneroden, K., Bowen, R., Rijal, K. R., Adhikari, B., & Ghimire, P. (2020). Effect of deworming on milk production in dairy cattle and buffaloes infected with gastrointestinal parasites in the Kavrepalanchowk district of central Nepal. VetRecord Open, 7(1), e000380. doi: 10.1136/vetreco-2019-000380
Song, Y., Endepols, S., Klemann, N., Richter, D., Matuschka, F. R., Shih, C. H., Nachman, M. W., & Kohn, M. H. (2011). Adaptive introgression of anticoagulant rodent poison resistance by hybridization between old world mice. Current biology: CB, 21(15), 1296-1301. doi: 10.1016/j.cub.2011.06.043
Ssuuna, J., Makundi, R. H., Isabirye, M., Sabuni, C. A., Babyesiza, W. S., & Mulungu, L. S. (2020). Rodent species composition, relative abundance, and habitat association in Mabira Central Forest Reserve, Uganda. Journal of Vertebrate Biology, 69(2), 20021. doi: 10.25225/jvb.20021
Stegniy, B. T., Mischenko, A. A., Zlotin, A. Z., Vovk, S. I., & Unkovskay, O. N. (2012). Mouse rodents - pests of livestock farms and adjacent agrobiocenoses. Kharkiv: NTMT, 236. (in Russian)
Stenseth, N. C. (2003). Mice, rats, and people: The bio-economics of agricultural rodent pests. Frontiers in Ecology and the Environment, 1(7). doi: 10.1890/1540- 9295(2003)001[0367:MRAPTB]2.0.CO;2
Suárez, O. V., & Cueto, G. R. (2018). Comparison of efficacy of second-generation anticoagulant rodenticides: Effect of active ingredients, type of formulation and commercial suppliers. Cogent Food & Agriculture, 4, 1, Article: 1525147. doi: 10.1080/23311932.2018.1525147
Swanepoel, L. H., Swanepoel, C. M., Brown, P. R., Eiseb, S. J., Goodman, S. M., Keith, M., Kirsten, F., Leirs, H., Mahlaba, T. A., Makundi, R. H., Malebane, P., von Maltitz, E. F., Massawe, A. W., Monadjem, A., Mulungu, L. S., Singleton, G. R., Taylor, P. J., Soarimalala, V., & Belmain, S. R. (2017). A systematic review of rodent pest research in Afro-Malagasy small-holder farming systems: Are we asking the right questions? PloS one, 12(3), e0174554. doi: 10.1371/journal.pone.0174554
Tanaka, K. D., Kawai, Y. K., Ikenaka, Y., Harunari, T., Tanikawa, T., Ando, S., wonMin, H., Okajima, F., Fujita, S., & Ishizuka, M. (2012). The genetic mechanisms of warfarin resistance in Rattus rattus found in the wild in Japan. Pesticide Biochemistry and Physiology, 103(2), 144-151. doi: 10.1016/j.pestbp.2012.04.011
Taylor, P. J., Downs, S., Monadjem, A., Eiseb, S. J., Mulungu, L. S., Massawe, A. W., Mahlaba, T. A., Kirsten, F., Maltitz, E. V., Malebane, P., Makundi, R. H., Lamb, J., & Belmain, S. R. (2012). Experimental treatment-control studies of ecologically based rodent management in Africa: balancing conservation and pest management. Wildlife Research, 39(1), 51-61. doi: 10.1071/WR11111
Walker, A. R. (2011). Eradication and control of livestock ticks: biological, economic and social perspectives. Parasitology, 138(8), 945-959. doi: 10.1017/S0031182011000709
Zavgorodniy, A. I., Stegniy, B. T., Paliy, A. P., Gorjeev, V. M., & Smirnov, A. M. (2013). Scientific and practical aspects of disinfection in veterinary medicine. Kharkiv: FOP Brovin, O.V., 222. ISBN 978-966-2445-59-6. (in Ukrainian)
Zuur, A. F., Ieno, E. N., & Elphick, C. S. (2010). A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol., 1(1), 3-14. doi: 10.1111/j.2041- 210X.2009.00001.x
Author Info
A.P. Paliy1*, N.V. Sumakova1, A.A. Antonіuk2, V.L. Behas2 and A.S. Panasenko32Polіsia National University, 7 Staryi Bulvar, Zhytomyr, 10008, Ukraine
3Sumy National Agrarian University, 160 Gerasim Kondratieva St, Sumy, 40021, Ukraine
Citation: Paliy, A.P., Sumakova, N.V., Antonіuk, A.A., Behas, V.L., Panasenko, A.S. (2021). Development and effectiveness of domestic bait in mouse-like rodents control. Ukrainian Journal of Ecology, 11 (2), 209-215.
Received: 25-Feb-2021 Accepted: 02-Apr-2021 Published: 30-Apr-2021, DOI: 10.15421/2021_101
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
